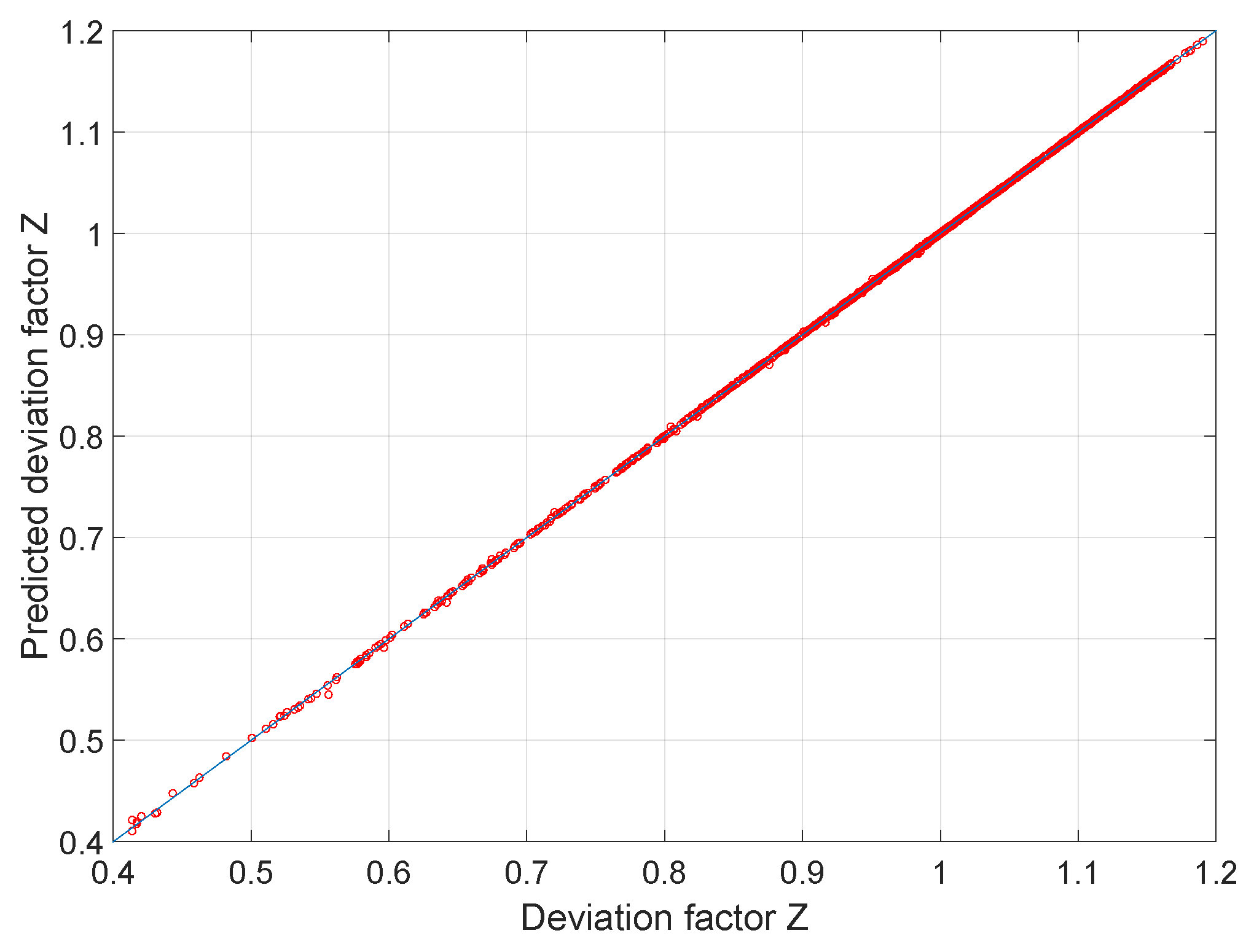
Compressibility factor for real gases
5 (749) · $ 19.00 · In stock
The compressibility factor Z for real gases can be rewritten

Solved Real gas effects can be expressed as departures from

Behaviour of Real Gases, PDF, Gases
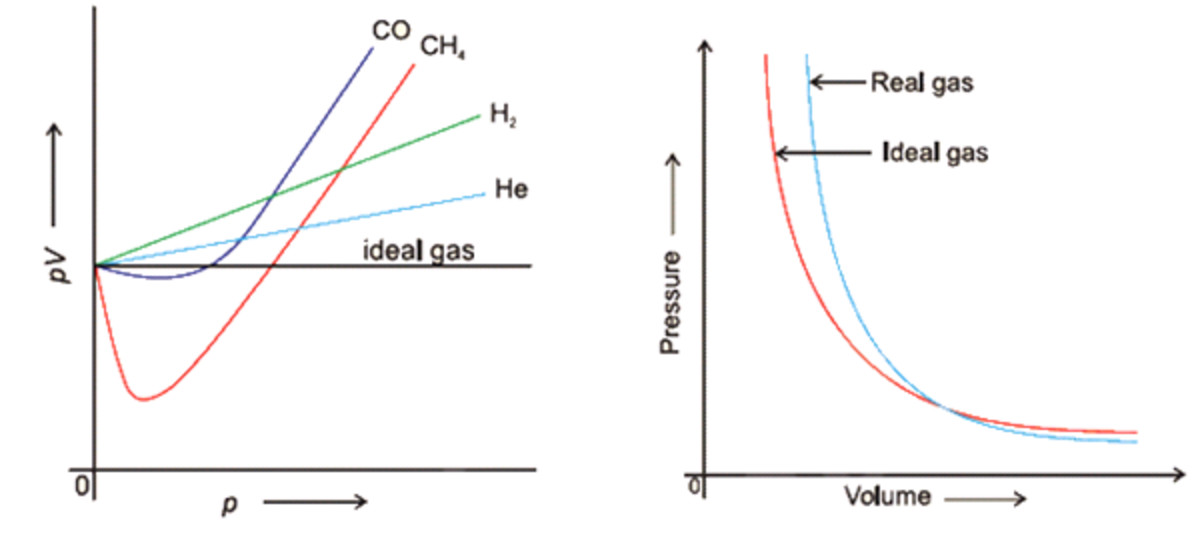
Real Gases - Chemistry, Class 11, States of Matter

Compressibility factor - Wikipedia

SOLVED: Show that P κ=1-P((∂ln z)/(∂ P))T for a real gas where κ is the isothermal compressibility.

What is the compressibility factor? What is its value an ideal gas

Chemistry Desk: Effect of Pressure

2. U 0.52, 0.68, 0.74 At low pressure, the comprensibility factor

Compressibility factor - Wikipedia

The given graph represents the variation of compressibility factor Z vs P for three gases A, B and C.Identify the incorrect statements.

For an ideal gas the compressibility factor is
Ideal gases and real gases are compressible or not compressible

Solved The graph of compressibility factor (Z)v/sP for 1 mol