Definition: The Universal or Ideal Gas Law describes the relationship between all four properties (pressure, volume, number of moles, and temperature) as well as a gas constant called “R.” NOTE: The Ideal Gas Constant “R” has constant a value of 0.0821 L.atm/mol.K Relation: The relation between pressure (P) volume (V), number of moles (n) and…

Ideal Gas Equation - Infinity Learn by Sri Chaitanya
Doretta Agostine

Combining Maxwell, Plank, and Bohr's Equations

6.3: Combining the Gas Laws: The Ideal Gas Equation and the General Gas Equation - Chemistry LibreTexts
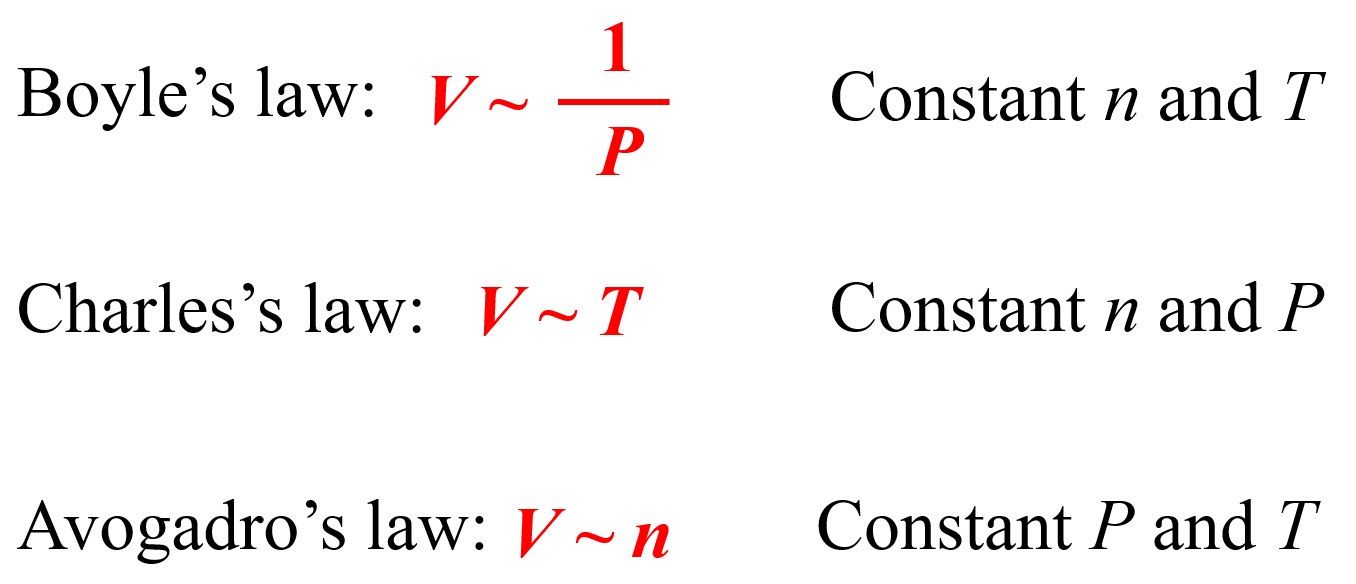
The Ideal Gas Law - Chemistry Steps
Ideal Gas Law - Wyzant Lessons
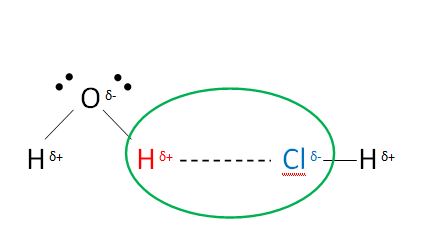
Intermolecular Forces of Attraction

Regents Chemistry--Physical Setting Power Pack Revised Edition by Albert S. Tarendash (Ebook) - Read free for 30 days

Universal Gas Law Study Guide - Inspirit Learning Inc
When do we use the universal gas constant R, and when do we use just the gas constant R? - Quora