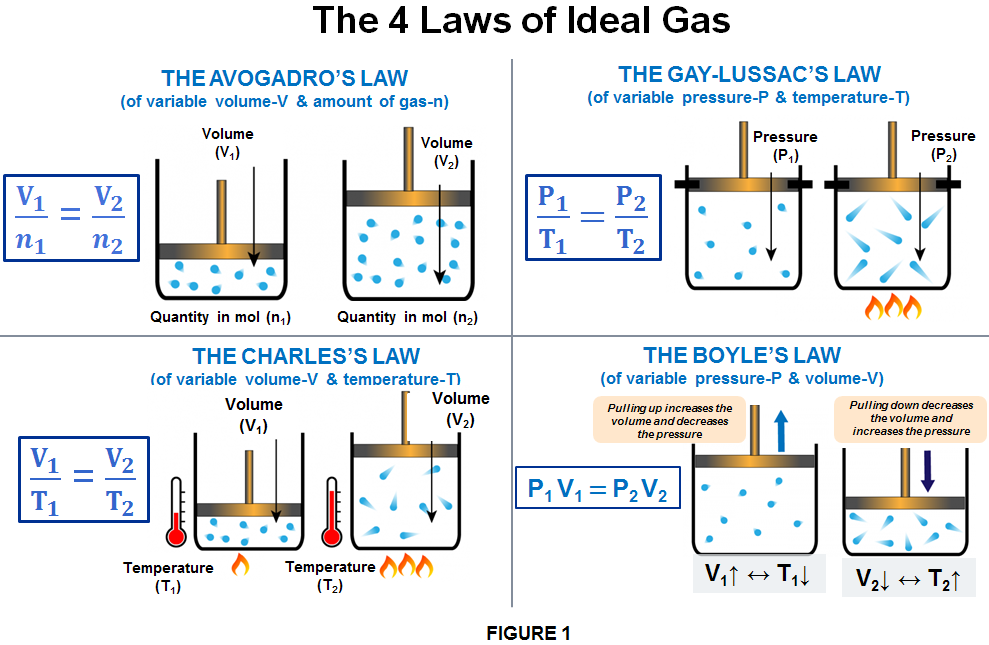
Ideal gas law, Definition, Formula, & Facts
4.5 (298) · $ 5.50 · In stock
Ideal gas law, relation between the pressure P, volume V, and temperature T of a gas in the limit of low pressures and high temperatures, such that the molecules of the gas move almost independently of each other. In such a case, all gases obey an equation of state known as the ideal gas law: PV =

/wp-content/uploads/2021/04/Avogad

Boyle's Law - Definition, Formula, Example

Calculating Pressure of an Ideal Gas - Derivation

Ideal–Universal Gas Law
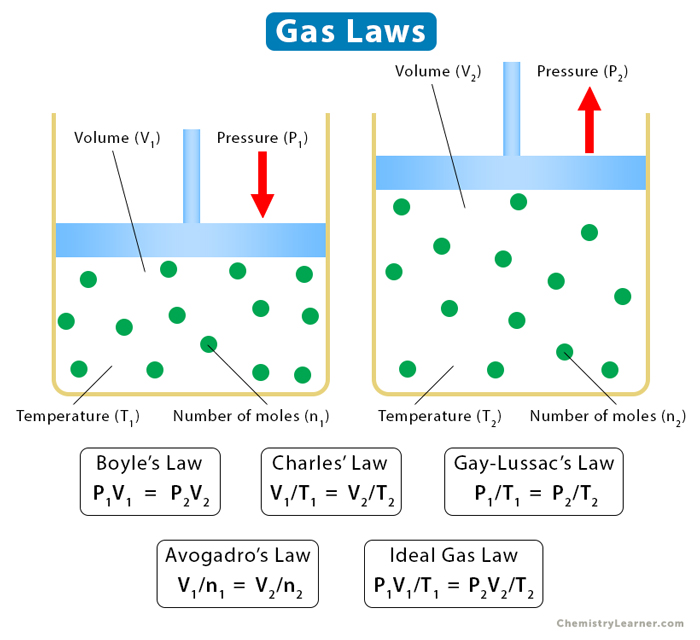
Gas Laws: Definition, List, Equations, and Problems
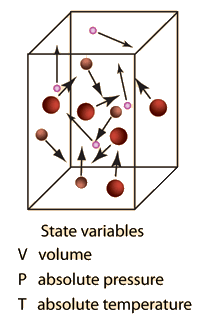
Ideal Gas Law
:max_bytes(150000):strip_icc()/149263439-56a12f093df78cf7726836ed.jpg)
Ideal Gas Law Example Problem

Ideal Gas Law

The Ideal Gas Law, Equation & Constant
:max_bytes(150000):strip_icc()/143058853-56a12f375f9b58b7d0bcdc3c.jpg)
Combined Gas Law Definition and Examples

Wake-up 1.Write the formula for Charles Law. 2.Write the formula for Boyle's Law. 3.Bromine gas has a pressure of mmHg. When it is dispensed into. - ppt download

Pressure, Temperature & Volume of a Gas