Absorption and Emission — Definition & Overview - Expii
4.6 (119) · $ 6.99 · In stock
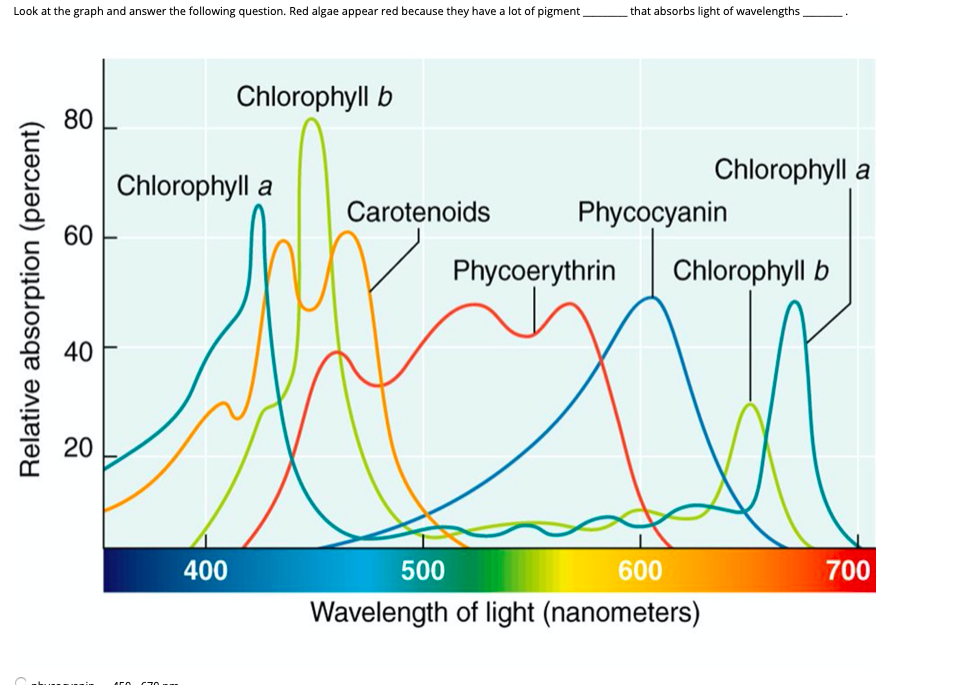
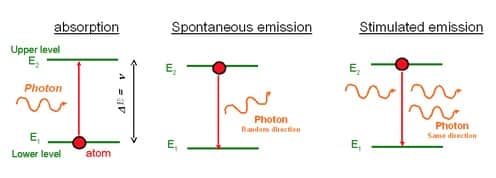
Absorption happens when an atom takes up energy. Emission happens when an atom gives off energy. Line spectra can be used to visualize these occurances.

Explain the difference between emission and absorption spectrum.

Emission and Absorption Spectra
What is Absorption and Emission? - StellarNet, Inc.

Absorption and Emission — Definition & Overview - Expii

Atomic Spectra — Overview & Application - Expii

Dr. Stanley Satz Revolutionized Alpha Therapy With Actinium-225 Discovery - PharmaFeatures

Neutrons — Structure & Properties - Expii

555 questions with answers in TRANSFER (PSYCHOLOGY)

Lesson Explainer: Emission and Absorption Spectra

Emission and Absorption Spectra

Lesson Explainer: Emission and Absorption Spectra
Emission & Absorption Spectrum: Emission,Absorption,Spectroscopy ,Line Spectrum

6.4: Emission and Absorbance Spectra - Chemistry LibreTexts

Continuous, Absorption and Emission Spectrum – Science Ready

Lesson Explainer: Emission and Absorption Spectra