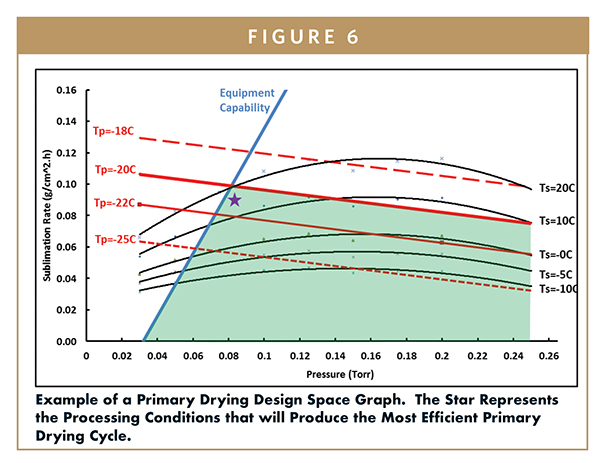
Example of a primary drying design space graph showing sublimation
4.6 (799) · $ 20.50 · In stock

Figure A1. Product temperature for 5.4 mL and 10.6 mL fill volumes

Practical Considerations for Freeze-Drying Process Design, Development and Scale-Up American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

Akhilesh BHAMBHANI, Merck & Co., Whitehouse Station

Sublimation interface location versus primary drying time – a 8.25 mm

The graphical design space for the primary drying phase of freeze Drying: Factors affecting the dried product layer resistance - ScienceDirect

Full article: Quality by design: optimization of a freeze-drying cycle via design space in case of heterogeneous drying behavior and influence of the freezing protocol

Recommended Best Practices for Lyophilization Validation-2021 Part

Recommended Best Practices for Lyophilization Validation—2021 Part I: Process Design and Modeling
Example of a primary drying design space graph showing sublimation

Ehab MOUSSA, Senior Scientist, PhD, AbbVie, Illinois
LYOPHILIZATION - Process Monitoring During Freeze-Drying