42g of N₂ react with excess of O₂ to produce NO. Amount of NO
4.7 (228) · $ 8.99 · In stock
Share your videos with friends, family, and the world

SOLVED: A mass of 6.45 g N2H4(g) reacts with excess oxygen. If 13.5 g NO2(g) is collected, what is the percent yield of the reaction? N2H4(g) + 3 O2(g) → 2 NO2(g) +

JP5198063B2 - Novel biocleavable linker - Google Patents

Limiting Reaction Calculations Practice Flashcards
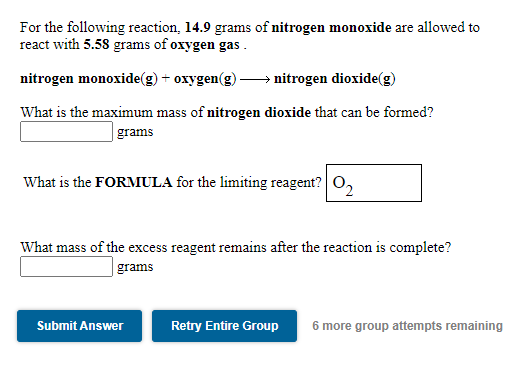
Solved For the following reaction. 14.9 grams of nitrogen

Limiting Reaction Calculations Practice Flashcards

27 g Al reacts completely with how many grams Oxygen.
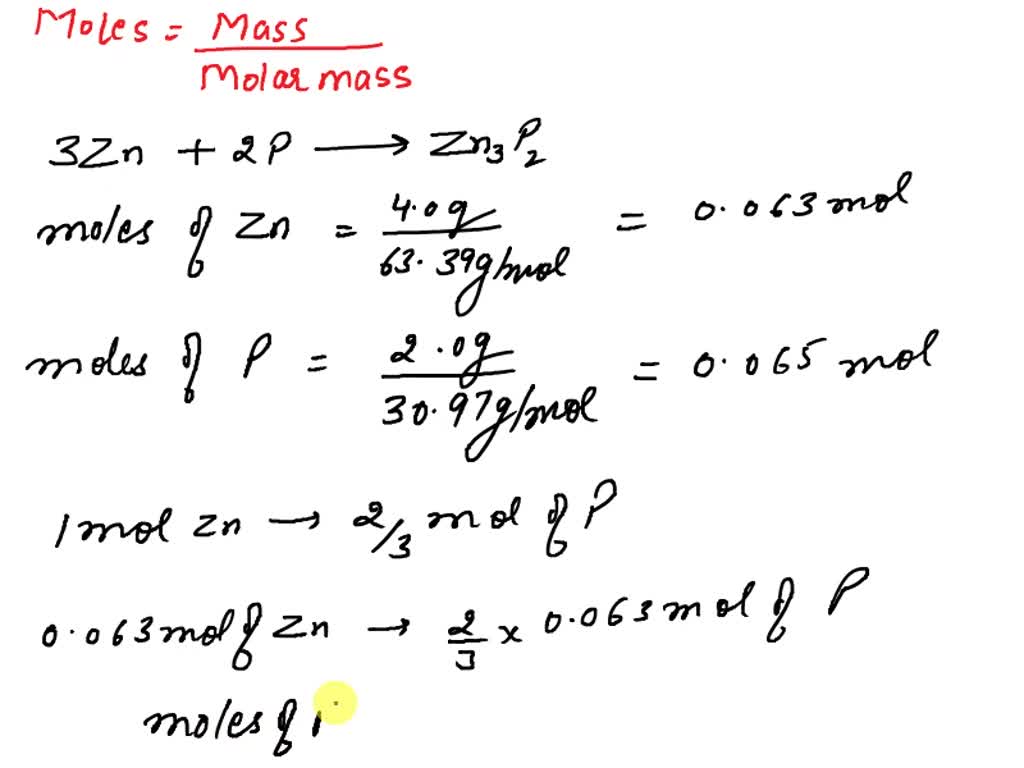
SOLVED: PQ-29. What amount of excess reagent remains when 4.0 g zinc reacts with 2.0 g phosphorus? 3Zn ZnP2 Molar mass Zn = 65.38 g/mol, P = 30.97 g/mol 0.70 g P (
When 14.5g of SO2 reacts with 21g of O2, what will be the theoretical yield and percentage yield of the reaction if the actual yield is 12g? - Quora
Solved] In the following reaction, oxygen is the excess reactant. SiCl 4
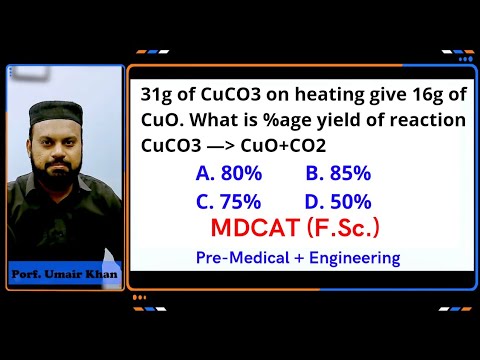
31g of CuCO3 on heating give 16g of CuO. what is %age yield of reaction. 80% 85% 75% 50%
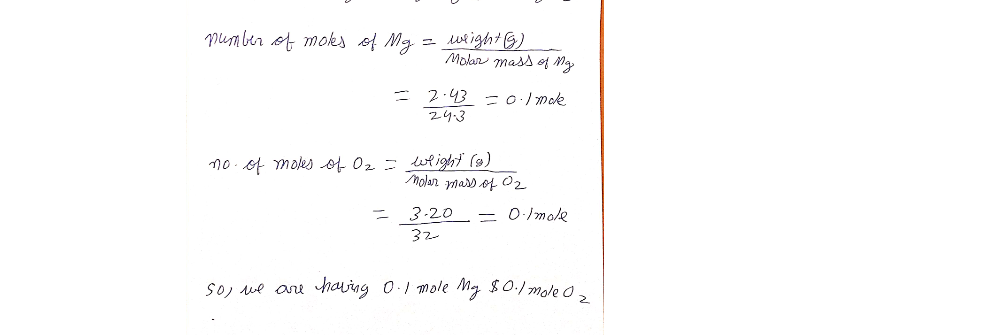
Answered: Suppose 2.43 g of magnesium is reacted…
N2 + O2 ---> 2NO, del H=+180 kJ /mol. Generally heat of reaction is written as 'x' kj 'per mole'. My question is in heat of reaction 'per mole' is of what (

UMAIR KHAN ACADEMY

Empirical formula of a hydrocarbon having 80% C and 20% of hydrogen is a.CH b.CH3 c.CH2 d.CH4 MDCAT












