- Home
- compressibility factor equation
- 1.7: Connecting the van der Waals and the viral equations: the Boyle temperature - Chemistry LibreTexts
1.7: Connecting the van der Waals and the viral equations: the Boyle temperature - Chemistry LibreTexts
5 (237) · $ 19.99 · In stock

Van der Waals Equation Practice Problems

Van der Waals Equation Practice Problems

1.7: Real Gases - Chemistry LibreTexts

For a van der Waal's gas, determine Boyle Temperature. [ given mathrm{a}=4.5 mathrm{atm} mathrm{L}^2mathrm{mol}^{-2}, mathrm{b}=0.9 mathrm{L} mathrm{mol}^{-1} and R=0.082 mathrm{L} mathrm{atm} mathrm{K}^{-1} mathrm{mol}^{-1}].609.8K6.09K273K60.98K

Boyle Temperature for Van der Waals, Berthelot and Dieterici, Unit 2, BPC Class

For a van der Waal's gas, determine Boyle Temperature. [ given mathrm{a}=4.5 mathrm{atm} mathrm{L}^2mathrm{mol}^{-2}, mathrm{b}=0.9 mathrm{L} mathrm{mol}^{-1} and R=0.082 mathrm{L} mathrm{atm} mathrm{K}^{-1} mathrm{mol}^{-1}].609.8K6.09K273K60.98K

Full PDF, PDF, Atomic Nucleus

Van Der Waals Equation of State - an overview
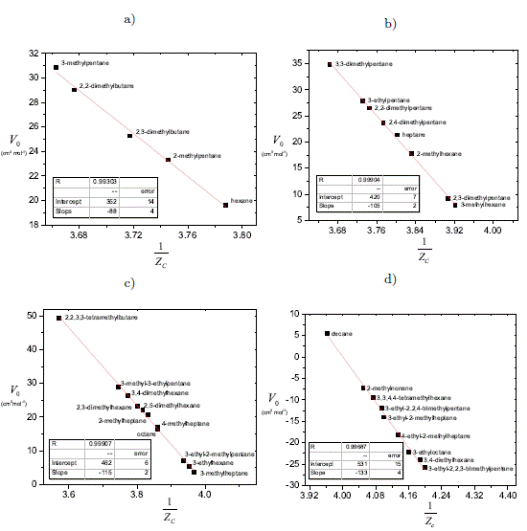
Critical Constants Correlation from van der Waals Equation












